ENT UK Revised Guidelines
ENT UK recently released their latest guidelines for potential Aerosol-Generating Procedures (AGPs) within the ENT Clinic, with a focus on advice for ENT Surgeons conducting upper-airway Endoscopy.
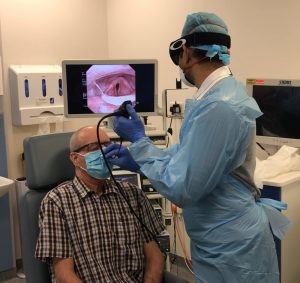
The Guidelines suggest that patients are advised to wear a mask during Nasendoscopy procedures, a problem that is solved using our Single-use SNAP, Endoscope Guide. This device maintains the Medical Device Regulation of the mask and is currently the only regulated and standardised method to do so, recently receiving UK Patent Application Number 2105626.2.
To see the full ENT UK Guidance, click here.
SNAP, Endoscope Guide
Using the SNAP device ensures safe passage of a Nasendoscopy through a Surgical Mask, creating an aperture for the Endoscope to pass through into the Nasal Cavity. Scientifically proven to reduce the prevalence of AGPs during examination, providing better safety and care both for the Patient & Clinician.
The device has been implemented throughout the United Kingdom and Ireland from which we are seeing huge success stories where the device is becoming standard practice.
If you would like to trial the multi-award-winning product in your Clinic, we are able to provide free samples, live demonstrations and have several training videos.
For more information on our SNAP, Endoscope Guide, please contact dtrmarketing@innoviamedical.com.