News » Gynaecology » Cervical Rotating Biopsy Punch Production Returns to Meet Increased Cancer Diagnosis
Cervical Rotating Biopsy Punch Production Returns to Meet Increased Cancer Diagnosis
Archive
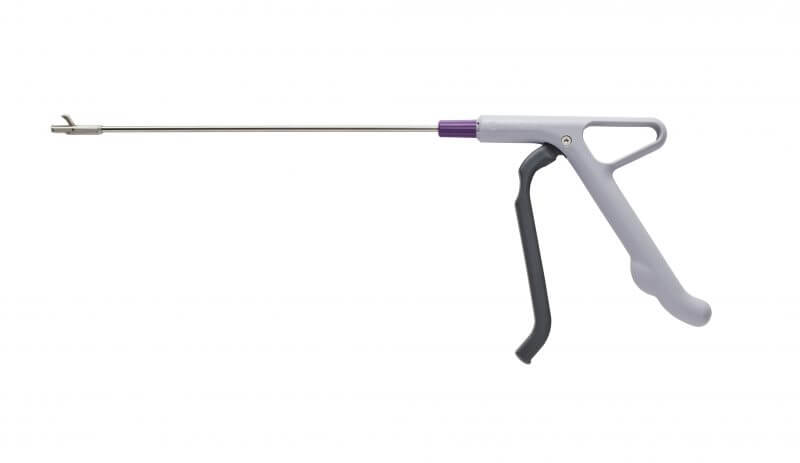
As more regular procedures return, the Innovia Medical family of companies are increasing their manufacturing capacity to ensure our customers have the products they require.
We are focusing on essential product lines to support urgent procedures such as cancer diagnosis, including our Cervical Rotating Biopsy Punch.
Our Single-use instrument eliminates cross contamination, inadequate samples and repeat procedures making it the instrument on choice for busy Colposcopy clinics dealing with the COVID-19 patient backlog. With around 2.1 million people currently waiting for breast, bowel or cervical screening in the UK[1] and Cervical cancer screening down 68% in the USA[2], the importance of a good biopsy is essential in reducing time to an accurate diagnosis. This has had a global impact with 42% of countries cancer treatment having been severely disrupted since the beginning of the COVID-19 pandemic[3].
What’s more our Biopsy Punch ensures a high-quality sample to be taken, even from a hardened cervix, leading to efficient Pathology laboratory results and quick diagnosis. This removes the dilemmas Clinics face when experiencing blunted reusable instruments that is; inadequate sampling, the impact in having to find an alternative handle or having to rearrange appointments to a different day resulting in patient recall.

For regular updates from across the Group, make sure you follow The Innovia Blog and our social media pages.
For more information on the Cervical Rotating Biopsy Punch and to discuss how else we can help you please contact marketing@dtrmedical.com.
References:
[1] https://scienceblog.cancerresearchuk.org/2020/06/01/impact-of-coronavirus-on-cancer-services-revealed-over-2-million-people-waiting-for-screening-tests-and-treatments/
[2] https://www.reuters.com/article/us-health-coronavirus-usa-screenings-exc/exclusive-u-s-medical-testing-cancer-screenings-plunge-during-coronavirus-outbreak-data-firm-analysis-idUSKCN22A0DY
[3] https://www.who.int/news-room/detail/01-06-2020-covid-19-significantly-impacts-health-services-for-noncommunicable-diseases